
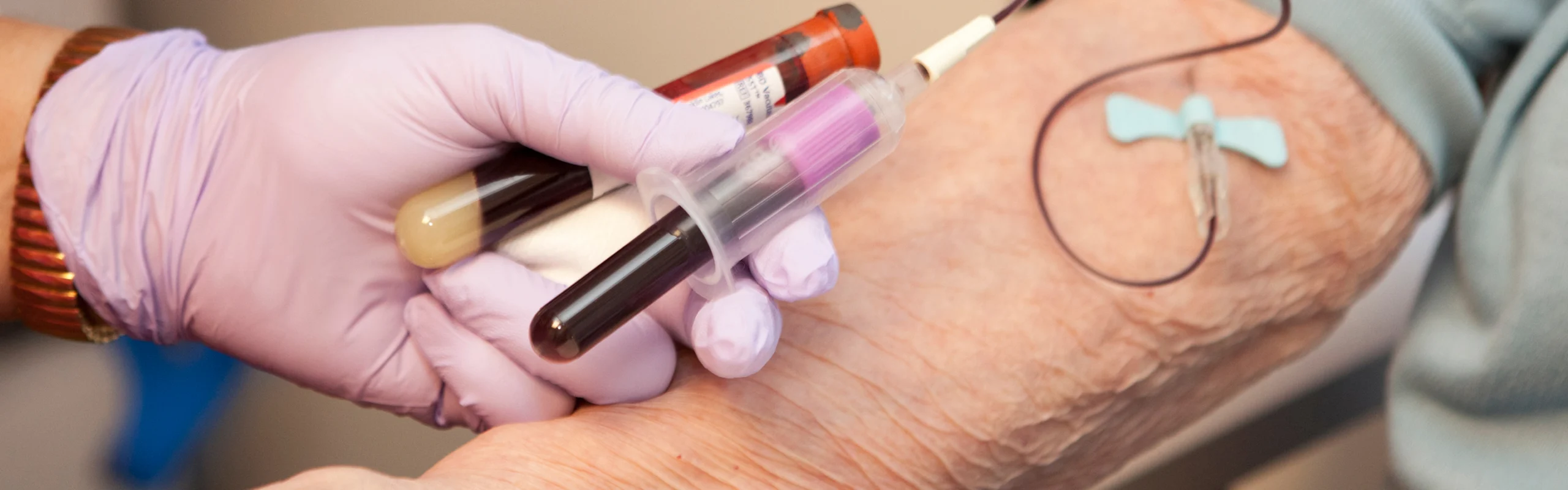
In the early 1980’s, Taiwanese hemophiliacs were infected with HIV by contaminated American-made medications. They or their surviving families filed claims for injuries they incurred due to those HIV-infections. Defendants are American pharmaceutical companies Bayer Corporation (formerly known as “Cutter”) and Baxter Healthcare who manufactured injected medications designed to stop hemophiliacs’ uncontrolled bleeding episodes.
Anti-hemophilia medications (“AHF”) were derived from paid donors whose plasma provided proteins that hemophiliacs needed to cause their blood to coagulate and stop bleeding. Those proteins, “Factor VIII” or “Factor IX,” were extracted from vats of plasma by a multi-step process that resulted in a freeze-dried powder. Thousands of plasma donations were pooled in those vats to make multiple products, including the AHF powder. AHF powder could then be dissolved in a saline solution and injected into infant, pediatric and adult hemophiliacs. Because AHF was ultimately injected, it was important that the plasma donors used to make AHF did not carry blood-borne diseases which would be transmitted to hemophiliacs when they infused AHF to stop bleeds.
In the late 1970’s and early 1980’s, U.S. law required the companies to use plasma from "normal, healthy donors" (U.S. Code of Federal Regulations (“CFR”) §§640.51 and 640.63) and to employ manufacturing practices to assure that blood products like AHF were “safe, pure, potent and effective” §606.140a. By 1975 it was known that individuals whose sexual activity included rectal intercourse with multiple partners had a high prevalence of hepatitis B and other sexually transmitted diseases, and it was recommended that these individuals “be advised to refrain from blood donations.” Szmuness, et al., On the Role of Sexual Behavior in the Spread of Hepatitis B Infection, Annals of Internal Medicine. October 1975; 489,494.]
Donors such as promiscuous urban “fast-track” gay males and prisoners who engaged in rectal intercourse, and intravenous drug users, were not considered to be “normal, healthy donors” and thus more likely to be high risk for transmitting blood-borne diseases such as HIV/AIDS and Hepatitis C. Notwithstanding, manufacturers sought these high-risk donors (1) since their life styles (or prison conditions) exposed them to diseases against which their bodies generated valuable antibodies which could be harvested and sold at high prices — antibody-rich general or disease-specific immune globulins were made from the same vats of pooled plasma used to make AHF. It was cost effective, although unsafe for hemophiliacs in the absence of a viral deactivation step, to make multiple products from the same vats of plasma.
1:Exhibit 8, Exhibit 9, Exhibit 10, Exhibit 11, Exhibit 12, Exhibit 13
AHF was first shipped to Taiwan from the U.S. in the late 1970s. By July of 1982, the Centers for Disease Control’s Morbidity and Mortality Weekly Report (2) was reporting that hemophiliacs were exhibiting symptoms of a disease previously observed in gay men, later termed “AIDS.” AHF manufacturers quietly met to discuss the “political, moral and liability” issues related to their having used plasma obtained from high-risk sources.
2:Exhibit 5, Exhibit 6
By December 1982, after injecting chimpanzees (3) with AHF and noting that they had developed “AIDS-like symptoms,” the manufacturers had strong proof that AHF was highly likely to transmit the agent that caused AIDS. Moreover, they had long known certain plasma-screening protocols would make AHF safer and that a viral-deactivation treatment was available. Bayer/Cutter’s medical consultant Dr. Lou Aledort of Mt. Sinai cautioned that once this product was licensed, it would be unethical to place a patient on other therapy” (emphasis supplied).
3: Exhibit 3, Exhibit 4
In 1983, Dr. Milton Mozen, head of Bayer/Cutter’s California research lab, wrote to its R&D director Dr. Sternberg and virologist, Dr. Robert Louie:
The recent concern about AIDS . . . should encourage a rapid review and approval of the submission [of an FDA license for heat-treated AHF]. . . [I]t is certainly logical that a heat-treated product . . . should be potentially safer than one not heated. Such a product should be made available . . . in as rapid a time frame as possible even without the final unequivocal demonstration of its freedom from hepatitis and/or AIDS.
However, in March of 1983, once Bayer/Cutter’s competitor, Baxter Healthcare, was granted its license for heat-treated AHF, Bayer/Cutter failed to heed the advice of Dr. Aledort and Dr. Mozen. Instead, Bayer/Cutter discouraged use of the new heat treated AHF, recommending use of AHF like Bayer’s that were supposedly subjected to heightened plasma screening. That is, keep using Bayer/Cutter’s un-heated AHF and don’t use Baxter’s heated AHF. (Exh. 65) Thus, heedless of the grave concern over an AIDS epidemic spreading amongst AHF patients, in their overseas-marketing materials (4) Bayer/Cutter suggested that a viral deactivation heat-treatment of AHF had not been shown to be effective. So instead of focusing on making AHF safer, it actively concealed its solicitation and use of high-risk blood donors (5) such as IV-drug users, prisoners, and promiscuous fast-track homosexuals; Bayer/Cutter dragged its feet in enacting safety measures such as viral deactivation; and it actively resisted product recalls. All this was done to protect profit (6) margins, with insufficient concern for hemophiliacs who were unknowingly infusing contaminated and deadly AHF medicine.
4:Exhibit 65, Exhibit 66, Exhibit 67, Exhibit 68
5: Exhibit 8, Exhibit 9, Exhibit 10, Exhibit 11, Exhibit 12, Exhibit 13
6: Exhibit 20, Exhibit 21, Exhibit 22, Exhibit 23
Bayer/Cutter maintained this marketing position critical of heat treating until Bayer/Cutter could market a heat-treated product of its own even after defendants’ heat-treated AHF was licensed. They began selling the safer AHF in the U.S. but continued to sell excess inventories of un-heated and HIV-contaminated AHF in foreign countries (7), including Taiwan with no plans to halt or recall contaminated AHF because they would lose money: “A major recall could deprive Cutter of up to $2 million [in] sales in the Far East . . .during 1984.”
7: Exhibit 20, Exhibit 21
Bayer/Cutter participated in a study to determine whether HIV survived the AHF manufacturing process. In May of 1984, it was reported that the study determined that it did. Likewise, the study tested whether HIV survived Bayer’s heat-treating process. The study determined that HIV did not survive Bayer’s heat-treating process. Later in 1984, the CDC MMWR published results of HIV testing in hemophiliacs who had been using un-heated AHF. 75% of frequent Factor VIII AHF users tested HIV positive. Thus, by mid 1984, Bayer knew its unheated product did not deactivate the HIV virus, that most hemophiliacs using it were testing HIV-positive, and that their heat-treatment process killed the HIV virus. Notwithstanding, it continued to sell and distribute its inventory of unheated AHF.
Likewise, by 1984, some countries were becoming aware of the AIDS risk from AHF. Bayer/Cutter’s 1985 Far East Marketing Plan (prepared in August 1984) lamented that the “luster” was gone from their AHF growth figures because sales to New Zealand “completely diminished” when “AIDS became an issue there.” Fortunately, as the plan advised, because hepatitis was prevalent in the Far East, “AIDS will not become a major issue amongst Asian hematologists during 1985.” So the company pushed even more contaminated AHF into the Far East, including Taiwan. Bayer/Cutter projected that “[the company would] have excess [AHF] in 1985 and [would] need to sell as much of it as possible, even at marginal prices. . .” But the plan warned “[h]ysteria over AIDS could reduce [AHF] sales. . .” Even so, the company had “no immediate plans to introduce Koate-HT. . .” instead intending to “dominate the [Taiwan] markets with low-cost standard Koate and Konyne” and to introduce heat-treated AHF only “if necessary to defend against AIDS.” To that end, it reassured Asian consumers that its unheated AHF was “not hazardous” and was the “same fine product we have supplied for years.”
Through June 1985, defendants actively depleted inventories of “this same fine product,” which by then had been banned in the U.S. U.S. Food and Drug Administration (“FDA”) head Dr. Harry Meyer told Cutter that it must quietly surrender its license for unheated AHF or it would be publicly revoked, a scenario he and the manufacturers wanted to avoid. Although AHF manufacturers, including Bayer/Cutter, “responded with a list of reasons why they had problems with the proposal, including the value of the inventory . . .” Meyer responded, “no one anywhere in the world should be allowed continued exposure to the HTLVIII virus.” Reluctantly, the company surrendered its license. (8)
8:Exhibit 18, Exhibit 19
Among the dangerous conduct Bayer/Cutter hid was its recall practice in Taiwan. In October 1983, it recalled 16 contaminated AHF lots shipped to both U.S. and international consignees, including Taiwan. The U.S. consignees were told all 16 lots were being recalled (9) because they contained plasma from a donor with AIDS, but the Taiwan consignee was only told that two lots were being recalled and then merely because a donor was suspected of having AIDS.
9:Exhibit 26, Exhibit 27
By April of 1984, Bayer/Cutter was training its international sales force to sell heat-treated AHF. Yet it delayed for over a year before sharing with overseas customers that heat-treated AHF was safer and non-heat treated AHF was a thing of the past. Finally, over 18 months after it began selling heat-treated AHF in the U.S., and five months after an international symposium had warned against using non-heat treated AHF, Bayer/Cutter notified its local rep in Taiwan to return any unheated AHF.
A few HIV-infected American hemophiliacs sued the AHF manufacturers in the mid 1980s. In the late 1980s and early 1990s, many more Americans sued, and their claims were settled in 1997. In re Factor VIII or IX Concentrate Blood Products Litigation, 159 F. 3d 1016 (7th Cir. 1998). More claims were litigated and/or settled separately around this time. See, e.g., Waage v. Cutter, 926 P. 2d 1145, (Alaska 1996); JKB v. Armour,et al, verdict 3-20-97; K.D.D. Smith v. Alpha, et al, No. 93-8088, Orleans Parish, LA, jury award 3-15-99, overturned on SOL, but settled.
Meanwhile, in early 1998, defendants negotiated an agreement through Taiwan’s Department of Health (“DOH”) and hemophiliac representatives to provide “humanitarian aid to hemophiliacs who contracted HIV.” In that negotiation, Bayer falsely claimed (10) that it was not at fault, and [had] won all relevant cases worldwide. Bayer representatives (11) who participated in the negotiations identified in discovery were Dr. Michael Strucksberg, a German resident who led the negotiation, and four Bayer employees from outside Taiwan: Horst Mueck of Germany, American attorneys, William Hammes and Thomas Kerr, and an individual named Paul Martin, whose current location has never been established. Bayer’s local counsel, attorney C.T. Lee, translated documents.
10:Exhibit 37, Exhibit 54
11:Exhibit 39, Exhibit 40
Dr. Strucksberg also prepared a news release which ran in Taiwan newspapers. A companion article was published in Taiwan’s United Daily News. Contrary to what they represented to the DOH and the press, in truth, defendants hadnot “won all relevant cases worldwide”: In March 1997, a jury had found Bayer negligent and awarded $2 million to plaintiffs. Bayer had also lost a summary-judgment motion, which led to a settlement.
Instead of disclosing these critical losses, Bayer asserted its innocence by claiming to have “won,” (12) a case where the plaintiff exclusively used another company’s AHF during the time period he became infected with HIV. Against this fraudulent backdrop, the defendants offered a nominal sum of $60,000 US, which they termed “humanitarian aid” for this “unavoidable tragedy,” to each Taiwanese hemophiliac.
12:Exhibit 42, Exhibit 42a
Crucial to the agreement was a “most-favored-nation” or “scale-up” clause which provided: “After having paid monetary compensations to some Claimants, if [the] Manufacturer decides to raise the compensation amount of paragraph 1 of this agreement or provide additional benefits in order to reach settlement with other Claimants regarding [the] Infection Incident, it shall also provide the same additional amount or additional benefits to Claimants who have already been paid.”
Consistent with the scale-up clause, Taiwan’s United Daily News reported on February 14, 1998, that Taiwan’s Minister of Health had said that the compensation/humanitarian aid “shall not be different from other countries.” Between the spring of 1998 and the end of 2002, Taiwanese plaintiffs signed a Chinese-language version of a humanitarian-aid agreement.
On May 22, 2003, the New York Times published an article entitled 2 Paths of Bayer Drug in 1980’s: Riskier Type Went Overseas, which revealed the defendants’ negligent, intentional, and fraudulent conduct. The article was republished in Taiwan newspapers.
Shortly thereafter, plaintiffs learned that defendants had paid other HIV-infected claimants more compensation. In May 2004 plaintiffs sent a letter to Bayer’s U.S. counsel requesting enforcement of the scale-up clause. The defendants refused, and plaintiffs timely sued in California.
The Taiwanese claims have been litigated as part of Multidistrict Litigation 986, as authorized by 28 U.S.C. §1407. Plaintiffs contend that the MDL court exceeded its authority in dismissing their tort claims on statute of limitations and forum non conveniens grounds and that after the Supreme Court’s ruling in Lexecon Inc. v. Milberg Weiss Bershad Hynes & Lerach, 523 U.S. 26, 118 S.Ct. 956, 140 L.Ed.2d 62 (1998), an MDL court’s jurisdiction does not extend to individualized case-specific issues
Since Lexecon, an MDL court’s authority is limited to consolidated or coordinated pretrial proceedings — matters for the benefit of the common litigation. Once those common-benefit matters are completed, the MDL court is statutorily mandated to request that a case be remanded for trial. Here, the MDL court actually did state that these cases appeared ready for remand to the transferor courts in California, and the plaintiffs agreed, but defendants requested that the MDL court decide statute of limitations issues first. Although plaintiffs stated that the statute of limitations and forum non conveniens issues should be decided by the California courts where the cases were originally filed, the MDL court proceeded with dismissing the tort and contract claims anyway. Plaintiffs contend that this was inconsistent with Lexecon’s ruling.
Taiwanese hemophiliacs have also asked the High Court to clarify anomalous lower federal court decisions where the doctrine of forum non conveniens has been used as a docket-clearing device to rid American courts of claims by foreign plaintiffs against American multinational companies through the broad flexibility granted the lower courts to weigh so-called public and private interest factors. Plaintiffs have asserted that a transfer to another jurisdiction is not appropriate where the inconvenience is merely shifted but not eliminated. Unless the moving party has shown that the chosen forum is vexatious and oppressive or inappropriate, dismissal on the basis of forum non conveniens is improper.
"Wisner Baum gave exceptional attention to all aspects of the case, detailed inquiry, and tenacious overview of all the information submitted. The paralegals are efficient and diligent. I was completely surprised to find an empathic personal message to take care of my own health during the challenging time of being a full-time caretaker.*"
In May of 2019, the jury in the case of Pilliod et al. v, Monsanto Company ordered the agrochemical giant to pay $2.055 billion in damages to the plaintiffs, Alva and Alberta Pilliod, a Bay Area couple in their 70s. R. Brent Wisner served as co-lead trial attorney for the Pilliods, delivering the opening and closing statements and cross-examining several of Monsanto’s experts. Wisner Baum managing shareholder, Michael Baum and attorney Pedram Esfandiary also served on the trial team in the Pilliod case.
The judge later reduced their award to $87M. Monsanto appealed the Pilliod’s verdict which the California Court of Appeal for the First Appellate District denied on August 9, 2021. Monsanto then requested the California Supreme Court review the appeal’s court decision, which the court denied on Nov. 17, 2021. Monsanto (Bayer) then submitted a petition for a writ of certiorari with the U.S. Supreme Court which SCOTUS denied on June 27, 2022, allowing the final judgment of $87M to remain intact.
$289.2 million jury verdict in Monsanto Roundup trial
Wisner Baum co-represented Dewayne “Lee” Johnson in the first Roundup cancer lawsuit to proceed to trial. On Aug. 10, 2018, a San Francisco jury ordered Monsanto to pay $39.25 million in compensatory damages and $250 million in punitive damages to Mr. Johnson, a former groundskeeper who alleged exposure to Monsanto’s herbicides caused him to develop terminal non-Hodgkin lymphoma.
Months after the jury verdict, the judge overseeing the trial reduced the punitive damages to $39.25 million. Mr. Johnson decided to accept the remittitur, bringing the adjusted amount awarded to Mr. Johnson $78.5 million.
Monsanto (Bayer) appealed the verdict and Johnson cross appealed. On July 20, 2020, the First Circuit Court of Appeals upheld the verdict against Monsanto but reduced Mr. Johnson’s award to $20.5 million. The company chose not to take the case to the U.S. Supreme Court, ending the litigation.
In 2016, Wisner Baum attorney Timothy A. Loranger and six other attorneys in the Plaintiffs’ Management Committee were able to secure a $265 million settlement for victims of the 2015 Amtrak 188 derailment in Philadelphia, one of the largest in the U.S. for 2016.